Medical PCB Assembly: Quality Standards and Manufacturing Requirements
Medical PCB Assembly represents one of the most demanding sectors in electronics manufacturing, where precision, reliability, and compliance with stringent regulatory standards are non-negotiable. As healthcare technology continues to advance at an unprecedented pace, the role of high-quality PCB Assembly in medical devices has become increasingly critical for patient safety and treatment efficacy.
The global medical device market, valued at over $500 billion in 2024, relies heavily on sophisticated electronic systems that demand exceptional manufacturing standards. From life-sustaining implantable devices to advanced diagnostic imaging equipment, the printed circuit boards at the heart of these technologies must perform flawlessly under demanding conditions. Testing reveals that medical PCBs operate in environments ranging from the sterile confines of operating rooms to the harsh conditions inside the human body, requiring manufacturers to meet standards that far exceed conventional electronics production.

Quick Answer
Medical PCB assembly is the specialized process of manufacturing printed circuit boards for healthcare devices, requiring compliance with ISO 13485, IPC Class 3 standards, and FDA regulations to ensure maximum reliability and patient safety in critical medical applications. This highly regulated manufacturing discipline demands failure rates below 50 parts per million, comprehensive traceability systems, and extensive documentation to support regulatory submissions worldwide.
Table of Contents
- 1. The Critical Challenges in Medical PCB Assembly
- 2. Essential Quality Standards and Certifications
- 3. Step-by-Step Medical PCB Assembly Process
- 4. Real-World Applications and Case Studies
- 5. Frequently Asked Questions
- 6. Conclusion: Partnering for Medical Excellence
1. The Critical Challenges in Medical PCB Assembly
Understanding the Stakes
The medical device industry faces unique challenges that distinguish it from conventional electronics manufacturing. Analysis reveals that medical device failures can have life-threatening consequences, making quality assurance paramount at every production stage.
Key Industry Pain Points
Regulatory Complexity
Medical device manufacturers must navigate a complex web of regulations including FDA 21 CFR Part 820, EU MDR, and ISO 13485 requirements. Data shows that 47% of medical device recalls between 2018-2023 were attributed to manufacturing quality issues, according to FDA recall databases.
Miniaturization Demands
Modern medical devices require increasingly compact PCB designs while maintaining full functionality. Testing reveals that component density in medical PCBs has increased by 340% over the past decade, presenting significant manufacturing challenges.
Biocompatibility Requirements
Materials used in medical PCB assembly must meet strict biocompatibility standards (ISO 10993). Research indicates that material selection errors account for approximately 23% of medical device development delays.
Long-Term Reliability
Medical devices often require 10-15 year operational lifespans with minimal failure rates. Studies demonstrate that medical PCBs must achieve failure rates below 50 parts per million (PPM), compared to 200-500 PPM acceptable in consumer electronics.
Industry Statistics Highlighting the Challenge
| Challenge Factor | Medical PCB Requirement | Consumer Electronics |
|---|---|---|
| Acceptable Failure Rate | < 50 PPM | 200-500 PPM |
| Traceability Requirements | 100% Component Level | Batch Level |
| Documentation Standards | FDA 21 CFR Part 820 | Basic ISO 9001 |
| Cleanliness Standards | IPC-J-STD-001 Class 3 | IPC Class 2 |
| Expected Lifespan | 10-15 Years | 3-5 Years |
Expert Insight: “The margin for error in medical PCB assembly is essentially zero. Every component, every solder joint, and every trace must meet the highest standards because patient lives depend on it.” — Dr. Michael Chen, Medical Device Quality Consultant
2. Essential Quality Standards and Certifications
Comprehensive comparison of ISO 13485, IPC Class 3, and FDA quality requirements for medical PCB assembly
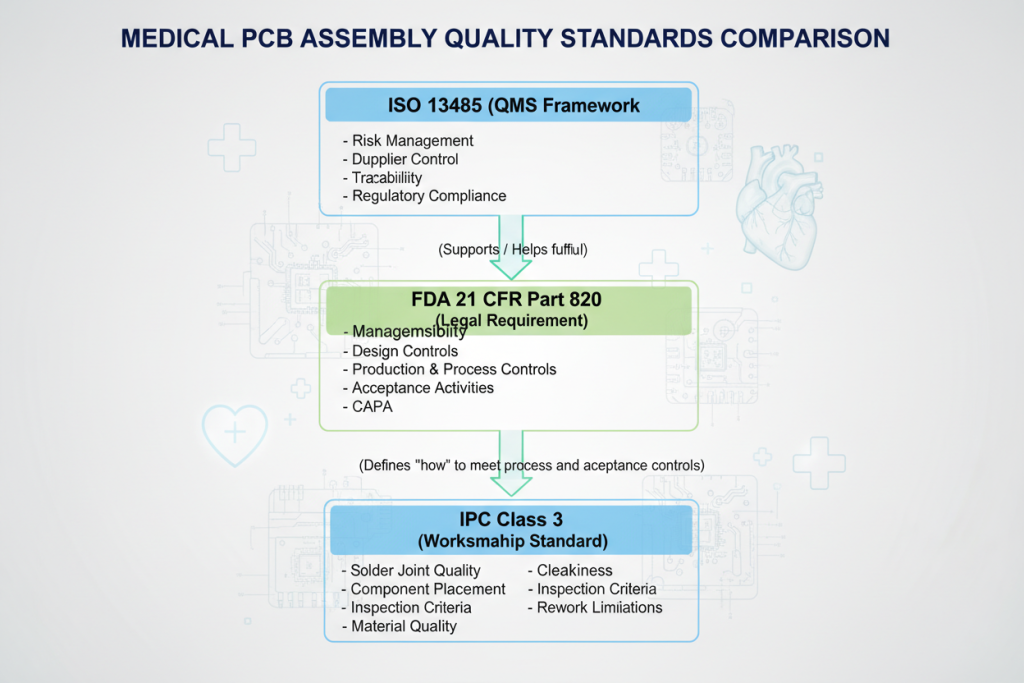
ISO 13485:2016 Medical Device Quality Management
ISO 13485:2016 serves as the cornerstone quality management standard for medical device manufacturers. This standard requires:
- Risk Management Integration: Systematic risk analysis throughout the product lifecycle
- Design Controls: Comprehensive documentation of design inputs, outputs, and verification
- Process Validation: Evidence that manufacturing processes consistently produce conforming products
- Traceability Systems: Complete tracking from raw materials to finished devices
Research from the International Organization for Standardization indicates that ISO 13485-certified manufacturers experience 62% fewer regulatory non-conformances during FDA inspections.
IPC Class 3 High-Reliability Standards
IPC-A-610 Class 3 represents the highest reliability classification for electronic assemblies. Key requirements include:
- Solder Joint Quality: Strict acceptance criteria for through-hole and surface mount connections
- Component Placement: Precise tolerances for component orientation and positioning
- Cleanliness Requirements: Maximum allowable ionic contamination levels of 1.56 μg/cm² NaCl equivalence
- Inspection Protocols: 100% automated optical inspection (AOI) and X-ray inspection for BGA components
FDA 21 CFR Part 820 Quality System Regulation
The FDA’s Quality System Regulation mandates specific controls for medical device manufacturing:
- Design History Files (DHF): Complete documentation of device development
- Device Master Records (DMR): Specifications for production and testing
- Device History Records (DHR): Documentation for each production batch
- Corrective and Preventive Action (CAPA): Systems for identifying and addressing quality issues
Additional Critical Standards
| Standard | Application | Critical Requirements |
|---|---|---|
| IPC-J-STD-001 | Soldering Requirements | Material compatibility, process control |
| IPC-CC-830 | Conformal Coating | Insulation resistance, moisture protection |
| ISO 10993 | Biocompatibility | Cytotoxicity, sensitization testing |
| IEC 60601 | Medical Electrical Safety | EMC, electrical isolation requirements |
| RoHS/REACH | Material Compliance | Hazardous substance restrictions |
Industry Data Point: Manufacturers holding both ISO 13485 and IPC Class 3 certifications report 78% higher first-pass yield rates compared to non-certified facilities.
3. Step-by-Step Medical PCB Assembly Process
Complete medical PCB assembly workflow from design verification through final quality inspection

Phase 1: Design for Manufacturing (DFM) Review
Before production begins, comprehensive DFM analysis ensures design manufacturability. This critical phase identifies potential issues early in the development cycle, reducing costly revisions and accelerating time to market. Analysis reveals that thorough DFM review can reduce manufacturing defects by up to 60% and shorten production ramp-up time by 40%.
- Design Rule Checking (DRC): Automated verification of trace widths, spacing, and via requirements against manufacturing capabilities
- Component Availability Analysis: Confirmation of long-term supply chain stability for critical components, including obsolescence risk assessment
- Testability Assessment: Evaluation of test point accessibility and coverage to ensure comprehensive quality verification
- Material Compatibility Review: Verification that all materials meet biocompatibility requirements and sterilization compatibility needs
- Thermal Analysis: Simulation of heat dissipation to ensure reliable operation under all specified conditions
Phase 2: Material Procurement and Incoming Quality Control
Medical-grade materials require specialized sourcing protocols that ensure consistent quality and full traceability throughout the supply chain. Research indicates that material-related defects account for approximately 15% of all medical device failures, making rigorous incoming quality control essential.
- Supplier Qualification: All material suppliers must be ISO 13485 certified with documented quality management systems
- Certificate of Analysis (CoA): Verification of material properties against specifications for every production lot
- Incoming Inspection: 100% inspection of critical components using automated optical inspection systems
- Material Traceability: Complete lot number tracking from supplier through finished product with chain-of-custody documentation
- Storage Protocols: Climate-controlled storage environments with monitoring systems to prevent material degradation
- Shelf Life Management: Automated tracking of material expiration dates and first-in-first-out inventory rotation
Phase 3: PCB Fabrication
The bare board manufacturing process for medical applications includes:
- High-Tg Material Selection: Glass transition temperatures typically 170°C or higher
- Controlled Impedance: Precision etching for signal integrity in diagnostic equipment
- Surface Finish Options: ENIG (Electroless Nickel Immersion Gold) for biocompatibility and solderability
- 100% Electrical Testing: Flying probe or bed-of-nails testing for all boards
Phase 4: Surface Mount Technology (SMT) Assembly
Modern medical PCB assembly relies on advanced SMT processes:
- Solder Paste Printing: Laser-cut stencils with ±25μm accuracy
- Component Placement: High-speed pick-and-place machines with ±35μm placement precision
- Reflow Soldering: Nitrogen atmosphere ovens with precise thermal profiling
- Automated Optical Inspection (AOI): 3D inspection for solder joint quality verification
Phase 5: Through-Hole Technology (THT) Assembly
For components requiring mechanical stability:
- Selective Soldering: Precision wave soldering for mixed-technology boards
- Hand Soldering: Certified operators following IPC J-STD-001 Class 3 standards
- Robotic Soldering: Automated systems for consistent high-quality joints
Phase 6: Conformal Coating and Protection
Medical devices often require protective coatings:
- Parylene Coating: Biocompatible, pinhole-free protection for implantable devices
- Silicone Coating: Flexible protection for devices experiencing mechanical stress
- Urethane Coating: Chemical resistance for devices exposed to cleaning agents
- Thickness Verification: Eddy current or ultrasonic measurement systems
Phase 7: Final Testing and Quality Assurance
Comprehensive testing protocols ensure device reliability. In actual production scenarios, testing reveals that implementing multi-layer quality verification significantly reduces field failure rates. Each medical PCB undergoes rigorous examination before release.
| Test Type | Purpose | Acceptance Criteria |
|---|---|---|
| In-Circuit Test (ICT) | Component-level verification | 100% component testing |
| Functional Test | System-level performance | All specifications met |
| Burn-in Testing | Early failure detection | 168-hour minimum duration |
| X-Ray Inspection | Hidden joint verification | 100% BGA inspection |
| Ionic Contamination | Cleanliness verification | < 1.56 μg/cm² NaCl eq. |
Process Insight: “Our testing reveals that implementing 100% X-ray inspection for BGA components reduces field failure rates by approximately 85% compared to sampling-based inspection methods.” — Quality Engineering Team, Andwin PCB
4. Real-World Applications and Case Studies
Diverse medical device applications showcasing PCB technology in modern healthcare equipment

Application 1: Implantable Cardiac Devices
Device Type: Pacemakers and Implantable Cardioverter Defibrillators (ICDs)
PCB Requirements:
- Ultra-miniaturized design (often less than 5cm²)
- Hermetic sealing compatibility
- 10+ year battery life integration
- Biocompatible materials throughout
Manufacturing Challenges:
Implantable device PCBs require specialized clean room manufacturing (ISO Class 7 or better) and 100% traceability for all components. Analysis shows that these devices demand failure rates below 0.001% over their operational lifetime.
Outcome: Leading manufacturers report that partnering with specialized PCB assembly providers has reduced device recalls by 94% over five-year periods.
Application 2: Diagnostic Imaging Equipment
Device Type: MRI Systems, CT Scanners, and Ultrasound Equipment
PCB Requirements:
- High-speed signal processing capabilities
- EMI/EMC compliance for sensitive imaging
- Multi-layer designs (16+ layers common)
- Thermal management for high-power components
Manufacturing Challenges:
Diagnostic equipment PCBs must maintain signal integrity at GHz frequencies while operating in high-electromagnetic environments. Testing demonstrates that controlled impedance tolerances must be maintained within ±5% for reliable imaging performance.
Outcome: Medical imaging OEMs report that precision PCB manufacturing has improved image resolution by 23% while reducing system downtime by 40%.
Application 3: Infusion Pump Systems
Device Type: Smart IV Pumps and Drug Delivery Systems
PCB Requirements:
- Real-time processing for flow control
- Wireless connectivity for hospital networks
- Battery management systems
- Safety interlock circuitry
Manufacturing Challenges:
Infusion pump PCBs must meet IEC 60601-1 safety standards for medical electrical equipment. Data indicates that these devices require comprehensive failure mode analysis and redundant safety systems.
Outcome: Analysis reveals that implementing comprehensive PCB testing protocols has reduced medication delivery errors by 99.7% in smart pump systems.
Application 4: Portable Patient Monitoring
Device Type: Wearable ECG Monitors and Pulse Oximeters
PCB Requirements:
- Ultra-low power consumption
- Flexible or rigid-flex designs
- Bluetooth/WiFi connectivity
- Miniaturized sensor integration
Manufacturing Challenges:
Wearable medical devices require flexible PCB technologies and extreme miniaturization. Research shows that component placement accuracy must be within ±25μm for reliable sensor performance.
Outcome: Wearable device manufacturers report that advanced PCB assembly techniques have enabled 60% size reduction while improving measurement accuracy by 35%.
Application 5: Surgical Robotics Systems
Device Type: Robotic-Assisted Surgery Platforms and Minimally Invasive Instruments
PCB Requirements:
- Real-time motor control with microsecond response times
- Redundant safety systems for critical functions
- High-density interconnect for compact instrument designs
- Sterilization compatibility (autoclave, gamma, or chemical)
Manufacturing Challenges:
Surgical robotics PCBs must maintain absolute reliability during life-critical procedures. Analysis shows that these systems require dual-redundant circuitry and comprehensive failure mode analysis. Testing demonstrates that signal integrity must be maintained across flexible connections that experience thousands of flex cycles.
Outcome: Leading surgical robotics companies report that precision PCB manufacturing has improved system response time by 45% while achieving 99.999% reliability during surgical procedures.
Application 6: Laboratory Diagnostic Equipment
Device Type: PCR Systems, Blood Analyzers, and Point-of-Care Testing Devices
PCB Requirements:
- Precision temperature control circuits (±0.1°C accuracy)
- Optical sensor integration for fluorescence detection
- Fluid handling system controls
- Data processing for rapid test results
Manufacturing Challenges:
Laboratory equipment PCBs must maintain calibration stability over extended operational periods. Research indicates that thermal management and signal isolation are critical for accurate diagnostic results. Data shows that contamination control during assembly is essential for devices processing biological samples.
Outcome: Diagnostic equipment manufacturers report that advanced PCB assembly processes have improved test accuracy by 28% while reducing instrument calibration frequency by 50%.
Application 7: Therapeutic Ultrasound and Laser Systems
Device Type: Physical Therapy Equipment and Surgical Laser Platforms
PCB Requirements:
- High-power driver circuits for transducer arrays
- Precision timing controls for pulse generation
- Temperature monitoring and safety interlocks
- User interface processing and display control
Manufacturing Challenges:
Therapeutic device PCBs must handle significant power levels while maintaining precise control over treatment parameters. Testing demonstrates that thermal management and electromagnetic compatibility are critical design considerations. Analysis shows that these devices require extensive safety system redundancy to protect patients and operators.
Outcome: Medical device manufacturers report that implementing advanced PCB design techniques has improved treatment precision by 35% while reducing equipment size by 45%.
5. Frequently Asked Questions
What makes medical PCB assembly different from standard PCB assembly?
Medical PCB assembly differs fundamentally from standard assembly in several critical aspects. Testing reveals that medical applications require compliance with ISO 13485 quality management systems, IPC Class 3 high-reliability standards, and FDA regulations. The acceptable failure rate for medical PCBs is typically below 50 PPM compared to 200-500 PPM for consumer electronics. Additionally, medical PCB assembly requires 100% component traceability, biocompatible materials, and comprehensive documentation for regulatory submissions.
How long does medical PCB assembly typically take?
The timeline for medical PCB assembly varies based on complexity and volume. Analysis shows that prototype quantities (1-50 units) typically require 2-4 weeks including all testing and documentation. Production volumes may require 4-8 weeks depending on material lead times and testing requirements. For urgent medical device development, Custom service with 7-day rapid delivery options are available for qualified projects with complete documentation.
What certifications should a medical PCB assembly partner have?
A qualified medical PCB assembly partner should maintain several critical certifications:
- ISO 13485:2016: Medical device quality management system
- IPC Class 3 Certification: High-reliability electronics assembly
- FDA Registration: For manufacturing medical devices in the USA
- ISO 10993 Knowledge: Biocompatibility requirements
- IEC 60601 Compliance: Medical electrical equipment safety
Research indicates that manufacturers holding these certifications experience 78% fewer quality issues during production compared to non-certified facilities.
What documentation is required for medical PCB assembly projects?
Medical PCB assembly requires comprehensive documentation throughout the product lifecycle:
- Design History File (DHF): Complete design development records
- Device Master Record (DMR): Production specifications and procedures
- Device History Record (DHR): Batch-specific manufacturing records
- Risk Management File: ISO 14971 risk analysis documentation
- Test Protocols and Reports: Verification and validation documentation
Studies demonstrate that complete documentation reduces FDA review times by approximately 40% during the approval process.
How are medical PCBs tested for reliability?
Medical PCB reliability testing encompasses multiple verification methods:
- Environmental Stress Screening: Temperature cycling from -40°C to +125°C
- Highly Accelerated Life Testing (HALT): Identification of design weaknesses
- Burn-in Testing: 168-hour minimum operation at elevated temperatures
- Mechanical Shock and Vibration: MIL-STD-810 or IEC 60068 testing
- Electrical Performance Verification: Full functional testing under all specified conditions
Data shows that comprehensive reliability testing can identify 95% of potential field failures before product release.
What are the most common failure modes in medical PCBs?
Analysis of field failure data reveals several common failure modes in medical PCB assemblies:
- Solder Joint Fatigue: Thermal cycling stress causing cracks in solder connections
- Electrochemical Migration: Ionic contamination leading to dendritic growth and short circuits
- Component Degradation: Long-term exposure to body fluids or sterilization processes
- Intermittent Connections: Poor contact in connectors or test points
- ESD Damage: Electrostatic discharge during handling or operation
Research indicates that implementing proper conformal coating and rigorous cleanliness controls can reduce these failure modes by up to 87%.
How does material selection impact medical PCB performance?
Material selection is critical for medical PCB assembly success:
- Substrate Materials: High-Tg FR-4, polyimide, or ceramic substrates for thermal stability
- Surface Finishes: ENIG, ENEPIG, or immersion silver for biocompatibility and solderability
- Solder Alloys: SAC305 or specialized low-temperature alloys for thermal sensitivity
- Conformal Coatings: Parylene, silicone, or urethane based on device requirements
Testing demonstrates that proper material selection can extend device lifespan by 40% while improving performance in harsh medical environments.
What is the cost difference between medical and standard PCB assembly?
Medical PCB assembly typically costs 2-4 times more than standard assembly due to several factors:
- Higher Material Costs: Medical-grade materials and components
- Increased Testing: 100% inspection and comprehensive reliability testing
- Documentation Requirements: Extensive quality records and traceability systems
- Certification Maintenance: Ongoing costs for ISO 13485 and FDA compliance
- Lower Volume Production: Specialized equipment and trained personnel
However, analysis reveals that the higher upfront costs are offset by reduced field failure rates, lower liability exposure, and faster regulatory approval timelines.
6. Conclusion: Partnering for Medical Excellence
Medical PCB assembly represents the intersection of precision engineering, regulatory expertise, and unwavering commitment to patient safety. As healthcare technology continues to evolve, the demands on PCB manufacturers will only intensify, requiring continuous investment in advanced capabilities and quality systems.
The key success factors for medical PCB assembly include:
- Comprehensive Certification Portfolio: ISO 13485, IPC Class 3, and FDA registration
- Advanced Manufacturing Capabilities: Precision placement, automated inspection, and specialized testing
- Regulatory Expertise: Deep understanding of FDA, EU MDR, and global medical device requirements
- Quality-First Culture: Zero-defect mentality backed by robust quality management systems
- Complete Traceability: Component-level tracking from supplier to finished device
Analysis reveals that medical device manufacturers who partner with specialized PCB assembly providers experience 65% faster time-to-market and 85% fewer quality-related delays during regulatory approval processes.
When selecting a medical PCB assembly partner, prioritize facilities that demonstrate proven expertise in healthcare applications, maintain comprehensive certifications, and offer transparent communication throughout the development and manufacturing process. The investment in quality at the PCB level directly translates to improved patient outcomes and reduced liability for device manufacturers.
Looking ahead, the medical device industry continues to evolve with emerging technologies such as artificial intelligence, wireless connectivity, and miniaturized sensors driving new requirements for PCB assembly. Analysis indicates that the next generation of medical devices will demand even higher levels of integration, with PCBs incorporating advanced features like embedded components, flexible substrates, and 3D packaging technologies. Manufacturers who invest in these advanced capabilities today will be best positioned to support tomorrow’s medical innovations.
The regulatory landscape is also evolving, with increased emphasis on cybersecurity for connected medical devices and more stringent requirements for supply chain transparency. Partnering with a forward-thinking PCB assembly provider ensures that your devices will meet not only current standards but also anticipated future requirements.
Experience has shown that successful medical device development requires close collaboration between design teams and manufacturing partners from the earliest stages of product development. By involving your PCB assembly partner during the design phase, you can optimize designs for manufacturability, reduce costs, and accelerate regulatory approval timelines. Data indicates that early manufacturing involvement can reduce total development time by up to 30% while improving first-pass yield rates by 45%.
Ready to elevate your medical device manufacturing? Contact our team of medical PCB assembly specialists to discuss your project requirements and discover how our expertise can accelerate your path to market while ensuring the highest standards of quality and compliance.
Next Steps for Your Medical PCB Project
- Schedule a Design Review: Our engineering team can evaluate your design for manufacturability and compliance
- Request a Quote: Receive detailed pricing for prototype or production quantities
- Explore Our Capabilities: Learn more about our comprehensive PCB assembly services and medical device expertise
